
Content

Dr. Yee Sook Cho of the Regenerative Medicine Research Center has established a new efficient method of commercializing reprogramming-related technologies and applying it to medical tests by developing a new small molecule (RSC133). This has the potential to improve the generation of induced pluripotent stem cells (iPSCs), similar to embryonic stem cells (ESCs), from human somatic cells.
RSC133, a newly identified small molecule, can be used to optimize the condition of reprogramming from patient’s own cells. As the technology is very promising to effectively regenerate tissue, Dr. Cho will continue to explore its potential molecule targets and applications for future stem cell based medical technologies.
Dr. Shinya Yamanaka (professor at Kyoto University, Japan), the 2012 Nobel Prize recipient for Physiology or Medicine, has first reported that somatic cells can be reprogrammed into a pluripotent state by ectopic expression of defined factors. This reprogramming technology offer several advantages over ESCs as therapeutic material, such as avoiding destruction of embryo and immune rejection following transplantation.
However, the use of viral transgenes, the slow reprogramming process, and low reprogramming efficiency impede potential applications of this technology.
Patient-specific iPSCs created from reprogramming technologies are highly-valued for their technical and economic value in various fields, including the development of customized treatments and new medicines.
In order to produce iPSCs with various levels of market-determined quality and to bring closer the date of commercialization, safe and effective reprogramming technologies should always have priority.
Dr. Cho’s team has identified RSC133, a new small molecule that increases reprogramming efficiency and they have characterized the functional role of RSC133 in the reprogramming process. (See Pictures 1 and 2)
As RSC133 enhances the growth of cells in the reprogramming process, regulates the expression of pluripotent marker genes and cell cycle inhibitory factors, and engages in epigenetic regulation, it demonstrates that it provides an intracellular environment favorable for reprogramming induction.
Moreover, when RSC133 has been added to a culture medium for pluripotent stem cells, including human ESCs and iPSCs, it has been demonstrated to inhibit differentiation and sustain an undifferentiated state effectively. Therefore, RSC133 may be used to develop techniques for the mass production of pluripotent stem cell cultures. (See Picture 2)
Dr. Cho has stated, “This research has shown the potential for improving the problems of current reprogramming technologies and bringing closer the commercialization of patient-customized treatments.”
This research was led by Dr. Yee Sook Cho and Dr. Jungwoon Lee of KRIBB and joined by Dr. Kyeong Lee of Dongguk University and Dr. Dongsup Kim of KAIST. It was also supported by the Biomedicine Technology Development Project of the Ministry of Education, Science and Technology (Minister Lee Jooho) and the National Research Foundation (President Seung-jong Lee) and by the NAP Cooperative Research Project by the Korea Research Council of Fundamental Science and Technology (President Kim Gun). A related achievement was also posted on the online version of ‘Angewandte Chemie International Edition’ (on 4th November, early view version), the world’s foremost journal in the field of chemistry. Domestic and international patents are pending.
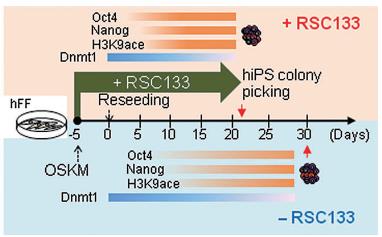
Picture 1.
▲RSC133 treatment during reprogramming process improved the efficiency and speed of reprogramming as well as the number of fully reprogrammed iPSCs. The time required for reprogramming was reduced (Green arrow). The timing of gene activation of the pluripotency markers (Oct4 and Nanog) and of histone (H3K9) acetylation is indicated by orange bars. The timing of gene repression of Dnmt1 is indicated by vanishing blue bars.
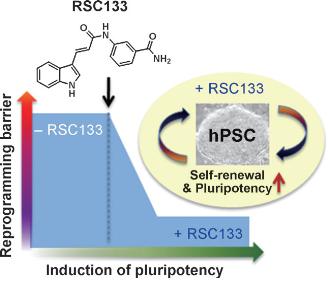
Picture 2.
▲RSC133 suppresses the reprogramming deterrent factors responsible for low reprogramming efficiency, and RSC133 is effective for maintaining and culturing undifferentiated pluripotent stem cells.
